Report: Salt-Vinega Power
A battery settles as one of the most important things of our life, because they are used often in modern society. Car engines, Digital Cameras, Laptops, IPhones, and Remote controls are some examples of materials that use a battery or batteries to operate it. In terminology, a battery is a combination of two or more electric cells, and one of its characteristics is that it converts the chemical energy to electric energy. Furthermore, the term battery is supposed to be for two electric cells, but in fact, we call it as a battery in common. It contains negative and positive terminals at the ends of it, so that it can receive and flow the electricity. Since a battery is concerned in chemical exercise, we need to be careful when we use it. This is because it can start a fire. In addition, as the amount of batteries being produced increases, it follows up with terrible pollution. I believe that if we find the way of producing the energy by some other way instead of using batteries, then it will decrease the pollution. As a consequence, I decided to pick a simple homemade battery for my science fair.
This simple homemade battery is the “Salt Vinegar Battery.” To someone, it will sound cool and fascinating, but at the same time they will think “Is that even possible?” As far as I am concerned, I think it is very possible to make a battery with salt and vinegar. To sum up, my testable question is this:
What salt concentration makes the best voltage?

For the materials, I will be required to prepare these items: 60ml of Vinegar, A small glass jar, Salt, Measuring spoons, Scale, Aluminum foil, Tape, Copper coated wire, Voltmeter and pencil. At this point, Aluminum foil and Copper coated wire are two different terminals in a battery. Aluminum foil will be the negative terminal and copper wire will be the positive terminal, even though both of them are positive elements due to the periodic table.
My topic wasn’t vinegar salt power at first, but it was “water salt power.” Reason that I changed the water to vinegar is that the vinegar’s characteristic is not only that it is an acetic acid but also there is salt present within it, so it tends to conduct the electricity more than water. Furthermore it means that if I use “Vinegar” instead of the water, it might offer me a better result.
Unlike other topics that my colleagues are doing, mine is very easy to clarify each of the variables. The independent variable of my experiment is the “Salt to Vinegar Ratio.” The dependent variable is the “Amount of Voltage.” Finally, my controlled variables are, “Amount of vinegar, same jar, same salt, same voltmeter, same alauminum foils, and same copper wire.”
Since this battery is easy and simple to make, it doesn’t take too much time. (Maybe 20 to 25 minutes) At first, you will be required to create a salt vinegar solution by mixing a small jar of vinegar with a teaspoon of salt. You must make sure the salt is dissolved into the vinegar, so that it can give you an exact result. Second, tape the aluminum foil on one side of the cup securely, and put it in the solution. As I mentioned before, this indicates the negative electrode (terminal). Third, tape a copper wire to the other side of the cup securely and also put it into the solution. This is the positive electrode of a battery. At this point, you need to check your electrodes whether they are taped tightly, because if they are not, it will be hard to measure the exact voltage. Since Aluminum foil and Copper wire are in the solution, we don’t need other things to conduct them, because they are conducting each other by liquid. Then, measure the dial on the voltmeter. At this point, you will be required to note how much voltage is flowing between the two electrodes. As soon as you record them, then put a spoon of vinegar again into the solution. (Measures the voltage again, once again, salt should be dissolved before connecting the voltmeter with a small glass jar.)
It will create voltage, because it contained all the supplements that a battery needed. As I mentioned, there are two electrodes inside of the beaker. Those two different electrodes will make electricity with the salt vinegar solution. This solution will help to flow chemical energy itself. Then, due to the chemical reaction it will create electric energy. Therefore, this experiment will be successfully finished
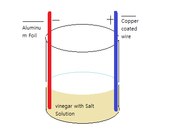
On your left side, there is a figure that I created and it tells you how my experiment will mostly look like. As you can see there is a beaker of salt vinegar solution with Aluminum foil (negative terminal) and Copper wire (positive terminal). My prediction about my experiment is as I put more salt into vinegar, so when the salt concentration is highest, its voltage will be the highest. This is because the more salt I put in a solution (salt and vinegar), more voltage it will create from there.
For
further prediction, salt vinegar battery has not only electrodes, but it also
will work as a secondary battery. (In a battery, there are two different types
in a large scale. One is called a primary battery, and the other one is called
a secondary battery. Primary battery is a battery that can’t be recharged
because chemical reactions are irreversible. On the other hand, Secondary
battery is a battery than can be recharged using electrical energy to reverse
the chemical reaction.) Let’s say, salt vinegar battery lost its voltage. At
that point, I can recharge the voltage by putting same amount of vinegar or
salt as before. Then, the voltage will recharge as it was before. .
On the top, I have told that this is working because there are electrodes that make conductivity to flow electricity in the solution. There is actually more to clarify and specific processes in terms of chemistry terminology. As soon as aluminum (Al) foil comes in contact with the surface of the salt vinegar solution, there happens a chemical action called “Oxidation,” Oxidation is the process of losing electrons, which are (-). At this point, Al becomes Al3+, it lost electrons itself. Then as the electrons approached the Copper coated (Cu) wire through the solution, the wire converts to Cu2+. At the end, it changed into Cu, because it gains the electrons. This process -gaining the electrons- is well known as “Reduction.” Anyway, this circulation will be continued constantly until it has lost its ability to pass through the conductivity, which rarely happens. You need to be aware of two characteristics of them. Not only Aluminum is a metal element, but Copper is a metal element. A combination of two metallic elements is called “Metallic Bonding.” To sum up, my battery is a result of the metallic bonding process that had a circulation of Oxidation and Reduction of the electrons constantly.
According to the Periodic Table, you can easily discover that neither of them are negative elements. If you realized this before I told you, then you might have a conversation with yourself about how it can work. This section is for resolving your curiosity. No one can deny the fact that both of them are positive metallic element due to the periodic table. However, Aluminum has greater tendency of losing electrons than Copper does. As a consequence, Al acts as the anion (AKA Negative Ion) while Cu loses electrons and becomes positive ion (cathode). Electrons flow from the Al to the Cu (- to +)
If salt-vinegar batteries become popular in society in the future, it will follow up with many benefits to us. First of all, it can contribute on the way of developing alternative energy. (However the problem is in the cost that are consuming for making an alternative energy used material.) Secondly, if we use it for now, we can actually decrease not only the tendency of pollution, but also the global warming. Therefore, we need to prefer to use alternative energy material like salt-vinegar battery, because it will be one of way to decrease the usage of perilous batteries.
On the top, I have told that this is working because there are electrodes that make conductivity to flow electricity in the solution. There is actually more to clarify and specific processes in terms of chemistry terminology. As soon as aluminum (Al) foil comes in contact with the surface of the salt vinegar solution, there happens a chemical action called “Oxidation,” Oxidation is the process of losing electrons, which are (-). At this point, Al becomes Al3+, it lost electrons itself. Then as the electrons approached the Copper coated (Cu) wire through the solution, the wire converts to Cu2+. At the end, it changed into Cu, because it gains the electrons. This process -gaining the electrons- is well known as “Reduction.” Anyway, this circulation will be continued constantly until it has lost its ability to pass through the conductivity, which rarely happens. You need to be aware of two characteristics of them. Not only Aluminum is a metal element, but Copper is a metal element. A combination of two metallic elements is called “Metallic Bonding.” To sum up, my battery is a result of the metallic bonding process that had a circulation of Oxidation and Reduction of the electrons constantly.
According to the Periodic Table, you can easily discover that neither of them are negative elements. If you realized this before I told you, then you might have a conversation with yourself about how it can work. This section is for resolving your curiosity. No one can deny the fact that both of them are positive metallic element due to the periodic table. However, Aluminum has greater tendency of losing electrons than Copper does. As a consequence, Al acts as the anion (AKA Negative Ion) while Cu loses electrons and becomes positive ion (cathode). Electrons flow from the Al to the Cu (- to +)
If salt-vinegar batteries become popular in society in the future, it will follow up with many benefits to us. First of all, it can contribute on the way of developing alternative energy. (However the problem is in the cost that are consuming for making an alternative energy used material.) Secondly, if we use it for now, we can actually decrease not only the tendency of pollution, but also the global warming. Therefore, we need to prefer to use alternative energy material like salt-vinegar battery, because it will be one of way to decrease the usage of perilous batteries.
Bibliography
Unknown, Salt-Water Batteries teacher's guide, Unknown dates, http://eburn.scripts.mit.edu/pened/doku.php/archive/lessons/battery/teachers.guide (April. 3rd. 2014)
Barry LeDrew, Jim Axford, Allan Carmichael, Kirsten Farquhar, Karen Morley, Jim Ried, and Malcolm Sneddon. B.C. SCIENCE PRBE 9. unkonwn Publication: Nelson. unknown copyright dates.
bexel_korea, Let's talk about a battery. (<- I translated in English) May. 15th. 2013. http://blog.naver.com/bexel_korea?Redirect=Log&logNo=40189068011
(April. 1st.2014) -> this website is written in Korean. i used this website, because i want to be aware of the working process of a battery clearly. (i can explain to you what is written in there.)
Unknown, Make a battery that works with air and saltwater Unknown dates, http://www.miniscience.com/projects/airbattery/, Mar. 27th. 2014
Barry LeDrew, Jim Axford, Allan Carmichael, Kirsten Farquhar, Karen Morley, Jim Ried, and Malcolm Sneddon. B.C. SCIENCE PRBE 9. unkonwn Publication: Nelson. unknown copyright dates.
bexel_korea, Let's talk about a battery. (<- I translated in English) May. 15th. 2013. http://blog.naver.com/bexel_korea?Redirect=Log&logNo=40189068011
(April. 1st.2014) -> this website is written in Korean. i used this website, because i want to be aware of the working process of a battery clearly. (i can explain to you what is written in there.)
Unknown, Make a battery that works with air and saltwater Unknown dates, http://www.miniscience.com/projects/airbattery/, Mar. 27th. 2014