What Is The Most Efficient Way To Extract Oil From Water?
 The non-polar molecules of oil being attracted to each other
The non-polar molecules of oil being attracted to each other
Science Report
Every year, millions of sea animals and birds such as seagulls die because of oil spills. The only way we can treat this situation is to find a method that best separates oil from water. Oil’s density is lower than water. It is about 800-900 kg/ m2. That is why it is easier to extract it from other liquid because it rises. This means that if you mixed coke and water, it is impossible to extract it. Oil is easier although it is still very hard to extract it.
Oil extraction is something that many scientists and environmentalists are working on. Extracting oil from water in a small cup is much easier than extracting it from the ocean. Although this is true, we could still apply the same methods of extracting oil from a cup to extracting it from a much bigger body of water.
There are some things in oil and water that make it hard oil and water to mix. Oil has certain molecules in it for us to be able to separate it from water. There is a simple rule that could explain this; similar liquids are able to dissolve in each other. Since oil and water are very different, they can’t. Oil’s density is about 800-900 kg/m3. Water is 1000 kg/m3. This causes oil to rise in water because it is not as dense.
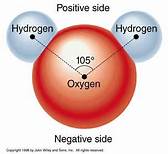
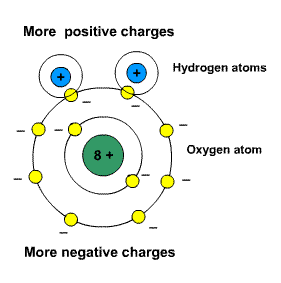
In water, there are polar molecules. This means that these molecules like to take sides.
They have slightly negative charges on one side and slightly positive charges on another.
The two oxygen atoms are clinging on to a single hydrogen atom. Polar molecules have no charge.
This makes them more individual.
On the other hand, oil has non-polar molecules; these are carbon atoms that have bonded with hydrogen atoms. This creates a hydrocarbon chain. This means that they are attracted more to each other so they don’t dissolve. These chains are like glue that keeps them together. This is what causes the oil to form a round shape on top of the water. They are not separated completely. Since water isn’t like that, it’s more individual molecules are easily separated.
If you shake a bottle mixed with water and oil, you will see that after some time, the oil will rise. This is because oil is slightly less dense than water. This is how lava lamps work; the oil is not as dense so it rises. They add salt since it’s denser than water, it makes the oil go down. Its all about density and the molecules in it.
This means that it would make it easier for people to separate it since it comes to the top. Also, the combination of its density and its non-polar molecules help and make it easier to extract it.
Every year, millions of sea animals and birds such as seagulls die because of oil spills. The only way we can treat this situation is to find a method that best separates oil from water. Oil’s density is lower than water. It is about 800-900 kg/ m2. That is why it is easier to extract it from other liquid because it rises. This means that if you mixed coke and water, it is impossible to extract it. Oil is easier although it is still very hard to extract it.
Oil extraction is something that many scientists and environmentalists are working on. Extracting oil from water in a small cup is much easier than extracting it from the ocean. Although this is true, we could still apply the same methods of extracting oil from a cup to extracting it from a much bigger body of water.
There are some things in oil and water that make it hard oil and water to mix. Oil has certain molecules in it for us to be able to separate it from water. There is a simple rule that could explain this; similar liquids are able to dissolve in each other. Since oil and water are very different, they can’t. Oil’s density is about 800-900 kg/m3. Water is 1000 kg/m3. This causes oil to rise in water because it is not as dense.
In water, there are polar molecules. This means that these molecules like to take sides.
They have slightly negative charges on one side and slightly positive charges on another.
The two oxygen atoms are clinging on to a single hydrogen atom. Polar molecules have no charge.
This makes them more individual.
On the other hand, oil has non-polar molecules; these are carbon atoms that have bonded with hydrogen atoms. This creates a hydrocarbon chain. This means that they are attracted more to each other so they don’t dissolve. These chains are like glue that keeps them together. This is what causes the oil to form a round shape on top of the water. They are not separated completely. Since water isn’t like that, it’s more individual molecules are easily separated.
If you shake a bottle mixed with water and oil, you will see that after some time, the oil will rise. This is because oil is slightly less dense than water. This is how lava lamps work; the oil is not as dense so it rises. They add salt since it’s denser than water, it makes the oil go down. Its all about density and the molecules in it.
This means that it would make it easier for people to separate it since it comes to the top. Also, the combination of its density and its non-polar molecules help and make it easier to extract it.